Groundbreaking advancements in vision restoration have emerged from an international clinical trial, co-led by notable experts such as José-Alain Sahel, M.D., from the UPMC Vision Institute, Daniel Palanker, Ph.D., from Stanford University, and Frank Holz, M.D., from the University of Bonn in Germany.
Clinical Trial Yields Remarkable Vision Improvements
In this study involving 32 participants over a year, an impressive 81% reported significant enhancements in their visual acuity. Furthermore, 84% of those surveyed indicated they utilized the artificial vision provided by the device at home for reading tasks.
On average, patients experienced a gain of 25 letters on a standard eye chart, which corresponds to an increase of approximately five lines. Notably, 81% of participants improved by at least 10 letters.
"This achievement marks a pivotal moment in vision restoration, as it's the first time such results have been documented in a significant patient cohort," stated Sahel, the study's senior author and chair of the Department of Ophthalmology at the University of Pittsburgh School of Medicine. "Over 80% of participants are now able to read letters and words, with some even reading full pages. This outcome surpasses our initial hopes when we embarked on this journey with Daniel Palanker 15 years ago."
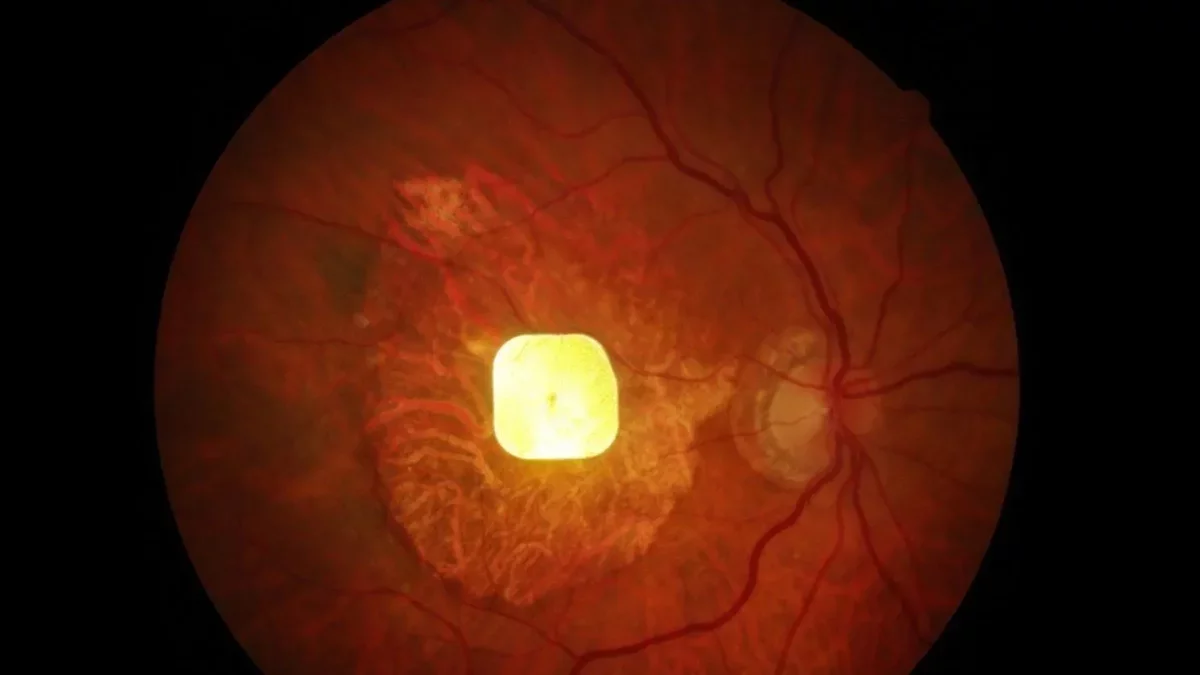
Understanding the PRIMA Retinal Implant
Age-related macular degeneration (AMD) leads to blurred central vision due to permanent damage to the retina's light-sensing cells. In healthy eyes, these photoreceptors capture light and convert it into electrical signals that are transmitted to the brain, forming images.
The PRIMA system, developed by Palanker, aims to replace damaged photoreceptors with a compact 2×2 mm wireless implant. This innovative device converts light into electrical signals, stimulating the remaining retinal cells. A camera integrated into specialized glasses captures images and transmits them to the implant using near-infrared light, which is then transformed into electrical pulses, aiding the brain in processing visual information. Users can customize zoom and contrast settings for enhanced clarity.
Comprehensive International Study
The PRIMAvera trial included 38 participants aged 60 and older across 17 medical centers in five European countries: France, Germany, Italy, the Netherlands, and the United Kingdom.
After a year of use, all side effects associated with the procedure had subsided, with most participants showing marked improvement in identifying letters on an eye chart. One participant even achieved a remarkable improvement of 59 letters, equivalent to 12 lines.
"Although we have yet to restore full 20/20 vision solely with the implant, our research at UPMC is focused on enhancing quality of life and potentially exceeding the legal blindness threshold," Sahel noted.
Future Prospects and Regulatory Approvals
In light of these promising results, the device manufacturer, Science Corporation, has initiated applications for clinical approval in both Europe and the United States. UPMC was the first U.S. institution to implant the PRIMA device in 2020, under the guidance of associate professor Joseph Martel, M.D.
Additional contributors to this study include institutions such as The University of Bonn, The Adolphe de Rothschild Foundation Hospital, Moorfields Eye Hospital in London, and the University of Rome Tor Vergata, among others. The research was funded by Science Corporation, based in Alameda, California.