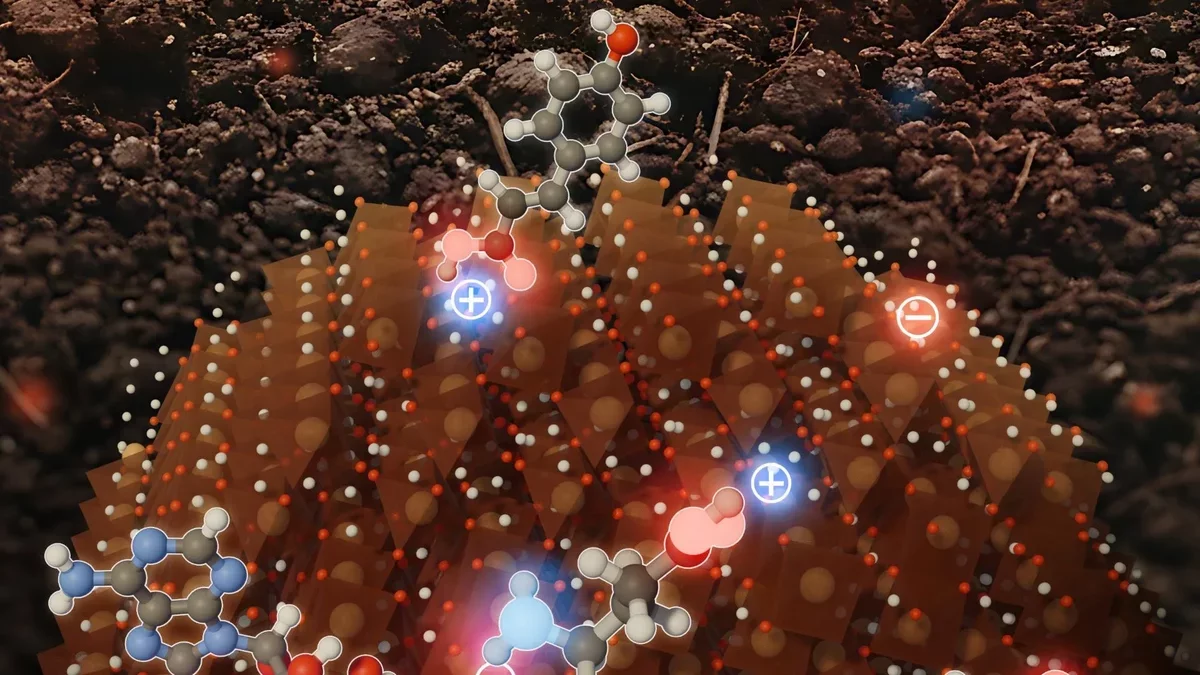
Engineers have made a fascinating discovery about ferrihydrite, a prevalent iron oxide mineral, revealing its complex strategies for capturing and retaining carbon. Instead of relying on a singular approach, this mineral utilizes a variety of chemical processes that enable it to bind different types of organic materials effectively.
Despite possessing an overall positive electrical charge, researchers have found that the surface of ferrihydrite is not uniform. It consists of tiny regions exhibiting both positive and negative charges. This uneven structure enhances ferrihydrite's ability to interact with carbon in ways that were previously underestimated. Alongside electrical attraction, the mineral forms both chemical and hydrogen bonds, establishing robust connections with organic molecules.
These combined mechanisms render iron oxide minerals remarkably versatile in their role as carbon binders. They can capture a diverse array of organic compounds and retain them for extended durations, sometimes spanning decades or even centuries. This process plays a crucial role in preventing carbon from re-entering the atmosphere as greenhouse gases, which contribute to climate change.
The research findings, published in the journal Environmental Science & Technology, provide an unprecedented insight into the surface chemistry of ferrihydrite, a vital element in understanding how soils store carbon.
"Iron oxide minerals are essential for the long-term preservation of organic carbon in soils and marine sediments," stated Ludmilla Aristilde from Northwestern University, who spearheaded the study. "The fate of organic carbon in the environment is closely tied to the global carbon cycle, which includes the transformation of organic matter into greenhouse gases. Therefore, comprehending how minerals trap organic matter is crucial, yet a quantitative assessment of how iron oxides bind various types of organic matter has been lacking."
Aristilde, a professor at Northwestern's McCormick School of Engineering, specializes in the behavior of organic materials within environmental systems. She is also associated with several prestigious institutes including the International Institute for Nanotechnology. The study's first author, Jiaxing Wang, along with second author Benjamin Barrios Cerda, are both postdoctoral associates in Aristilde's lab.
Soil: A Major Carbon Reservoir
Soil is estimated to store around 2,500 billion tons of carbon, making it one of the largest carbon reservoirs on Earth, second only to the oceans. Despite its significance, scientists are still exploring the precise mechanisms that enable soil to sequester carbon from the active carbon cycle.
Aristilde and her team have dedicated years to investigating how minerals and soil microbes affect whether carbon remains sequestered or is released back into the atmosphere. Their prior studies focused on how clay minerals bind organic matter and how certain microbes preferentially convert specific organic compounds into carbon dioxide.
In this recent research, the focus shifted to iron oxide minerals, which are associated with over a third of the organic carbon present in soils. The team concentrated on ferrihydrite, commonly found in proximity to plant roots and in soils or sediments abundant in organic material. While ferrihydrite typically exhibits a positive charge in environmental conditions, it can bind organic compounds with negative, positive, or neutral charges.
Understanding Molecular Interactions with Iron Minerals
To comprehend how ferrihydrite interacts with such a wide variety of compounds, the researchers employed high-resolution molecular modeling and atomic force microscopy to scrutinize the mineral's surface. They confirmed that, despite its overall positive charge, the surface comprises a blend of positive and negative regions, which clarifies why ferrihydrite can attract negatively charged substances like phosphate, as well as positively charged metal ions.
"It is well established that ferrihydrite carries a positive charge in relevant environmental conditions," Aristilde noted. "This has led to the assumption that only negatively charged compounds can bind to these minerals, but our findings demonstrate that these minerals can indeed bind compounds with both negative and positive charges. Our research illustrates that the overall positive charge of the mineral arises from the combination of both types of charges distributed across its surface."
After mapping the surface charges, the team examined how various organic molecules interact with ferrihydrite. They exposed the mineral to compounds commonly found in soil, such as amino acids, plant acids, sugars, and ribonucleotides. The researchers quantified how much of each compound adhered to the mineral and utilized infrared spectroscopy to analyze the attachment of the molecules.
Beyond Simple Attraction
The experiments unveiled that ferrihydrite binds organic molecules through multiple distinct pathways. Positively charged amino acids attach to negatively charged areas of the mineral, while negatively charged amino acids bind to positively charged regions. Certain compounds, like ribonucleotides, are initially attracted by electrical forces but subsequently form stronger chemical bonds with iron atoms. Sugars, which have a weaker binding affinity, attach through hydrogen bonding.
"Our collective findings provide a quantitative basis for developing a framework to understand the mechanisms driving mineral-organic associations involving iron oxides in the long-term preservation of organic matter," Aristilde stated. "These associations may clarify why some organic molecules remain safeguarded in soils while others are more susceptible to degradation by microbes."
The researchers plan to investigate the fate of organic molecules after they bind to mineral surfaces. Some may be transformed into compounds that microbes can further decompose, while others could become even more resistant to breakdown.
The study, titled "Surface charge heterogeneity and mechanisms of organic binding modes on an iron oxyhydroxide," received support from the U.S. Department of Energy and the International Institute for Nanotechnology.