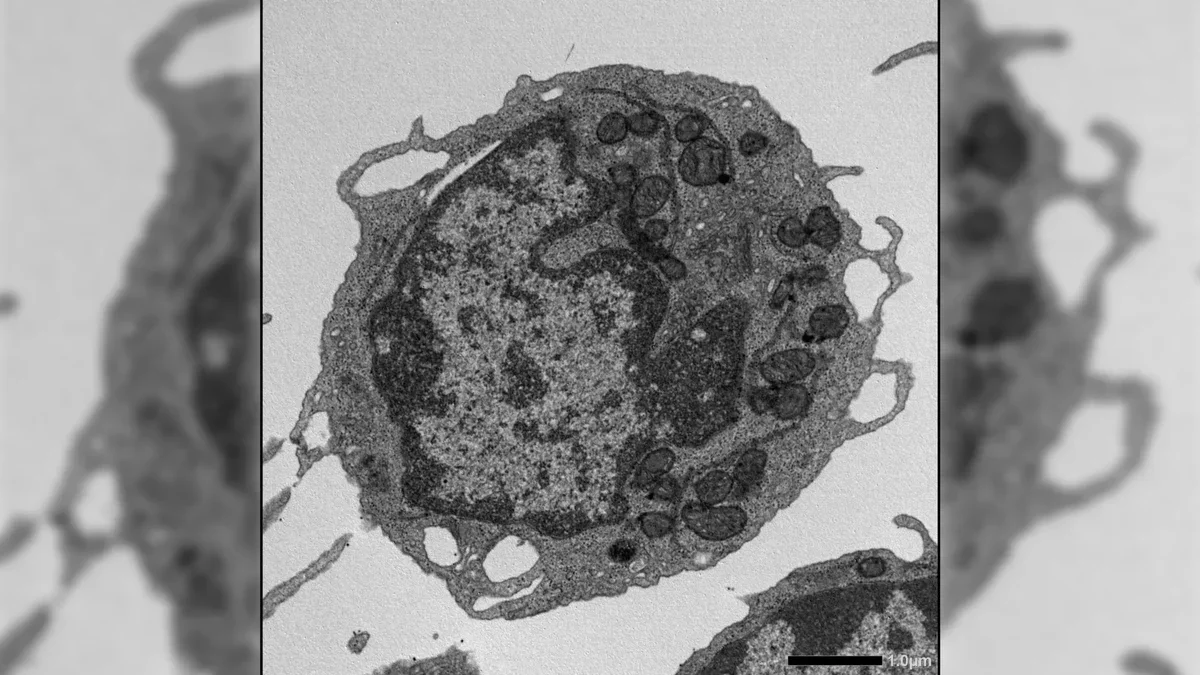
Research has unveiled several factors contributing to the decline of hematopoietic stem cells (HSCs), including accumulated cellular damage, changes in gene activity, and chronic low-level inflammation. However, the intricate interplay of these stresses affecting HSC function has remained elusive.
Exploring a Critical Aging Pathway
To delve deeper into this phenomenon, a collaborative team from The University of Tokyo and St. Jude Children's Research Hospital investigated how age-related stress impacts HSCs. They concentrated on the RIPK3-MLKL signaling pathway, traditionally associated with necroptosis, a form of programmed cell death.
Dr. Masayuki Yamashita led the study, which included contributions from Dr. Atsushi Iwama and Dr. Yuta Yamada. The research was published in Volume 17 of Nature Communications on April 6, 2026.
Unexpected Findings on MLKL
The researchers encountered an intriguing observation: HSCs from MLKL-knockout mice treated with 5-fluorouracil showed significantly fewer aging-related functional changes, despite no difference in HSC death rates. This prompted an investigation into whether MLKL could influence stem cell aging without inducing cell death.
Examining the Mechanism
To test this hypothesis, the team utilized various genetically modified mice and specialized reporter mice to monitor MLKL activation. They subjected the mice to stress conditions mimicking aging, such as inflammation and replication stress, and assessed HSC functionality primarily through bone marrow transplantation.
Advanced techniques, including flow cytometry and high-resolution imaging, provided comprehensive insights into MLKL's impact on HSCs.
Mitochondrial Damage Without Cell Death
The study revealed a novel role for MLKL in stem cell aging. Although typically linked to cell death, MLKL activation in HSCs did not increase cell mortality. Instead, it transiently relocated to the mitochondria, where it induced damage by lowering membrane potential and impairing energy production. This damage contributed to aging-related declines in HSC function.
Preserving Stem Cell Function
When MLKL was removed or inactivated, the adverse effects were considerably mitigated. HSCs without MLKL maintained their regenerative capacity and produced healthier immune cells, even under stress or in older animals. These improvements occurred without significant changes in gene expression, suggesting that MLKL influences aging through structural cellular changes rather than direct genetic regulation.
Future Implications for Aging Research
The findings illuminate a shared pathway linking various cellular stresses to mitochondrial damage and stem cell aging. By highlighting MLKL's role, this research opens avenues for developing therapies aimed at preserving HSC function, potentially enhancing recovery for patients undergoing treatments like chemotherapy.
Dr. Yamashita notes, "This research could pave the way for new mitochondrial-protective or necroptosis-modulating drugs, ultimately transforming how we approach age-related decline in the blood and immune systems."