Current bone implants often rely on autografts--using the patient's own bone--or metal and ceramic materials. Autografts necessitate an extra surgical procedure to harvest bone tissue, which can lengthen recovery time and elevate surgical risks. Metal implants, while durable, can pose challenges due to their rigidity compared to natural bone, potentially leading to loosening and reduced long-term stability.
Creating Biologically-Compatible Bone Implants
Bone is a complex structure, featuring numerous microscopic tunnels and cavities crucial for its strength and functionality. "Incorporating biological elements into the healing process is essential," states Xiao-Hua Qin, a Professor of Biomaterials Engineering at ETH Zurich. Effective bone repair relies on various cell types migrating into the implant and collaborating to regenerate tissue.
To better align with this biological intricacy, Qin and his team, alongside ETH Professor Ralph Müller, have designed a novel hydrogel intended for future bone implants. This soft material, akin to jelly, gradually dissolves within the body, potentially allowing for personalized implants tailored to individual needs. Their research findings have been published in Advanced Materials.
Mirroring the Body's Natural Repair Mechanism
Upon a bone fracture, the body does not immediately produce hard tissue. Instead, it forms a soft, permeable scaffold. Initially, a hematoma develops at the fracture site, facilitating the movement of immune and repair cells while supplying necessary nutrients. A network of fibrin binds these cells together, which eventually transforms into solid bone over time.
The innovative hydrogel mimics this initial healing stage, comprising 97% water and 3% biocompatible polymer. To regulate its hardening process, researchers incorporated two specialized molecules: one for linking polymer chains and another that activates upon light exposure, initiating solidification.
Wanwan Qiu, a former doctoral student, developed the linking molecule for this purpose. "It allows for rapid structuring of hydrogels at the sub-micrometer level," she explains. When targeted laser pulses hit the material, polymer chains bond instantly, forming a solid structure, while areas shielded from the laser remain soft for later removal.
Record-Setting Laser Printing at Nanoscale
This method enables the team to shape the hydrogel with remarkable precision, achieving structures as small as 500 nanometers. "Hydrogels can be challenging to mold," notes Professor Qin. "Our new connecting molecule allows us to structure the hydrogel stably and finely while achieving high printing speeds of up to 400 millimeters per second--a new world record."
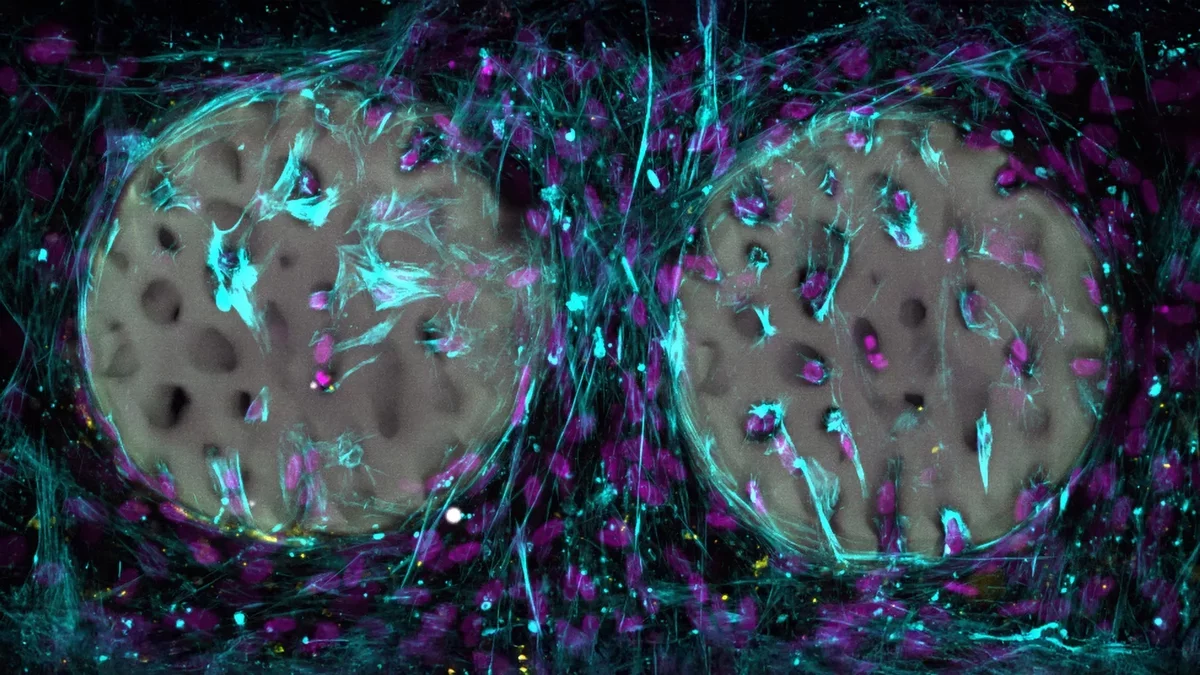
In their experiments, the researchers successfully created intricate hydrogel structures modeled after actual bone. Utilizing medical imaging, they replicated the delicate trabecular lattice that imparts internal strength to bones.
Promising Laboratory Results
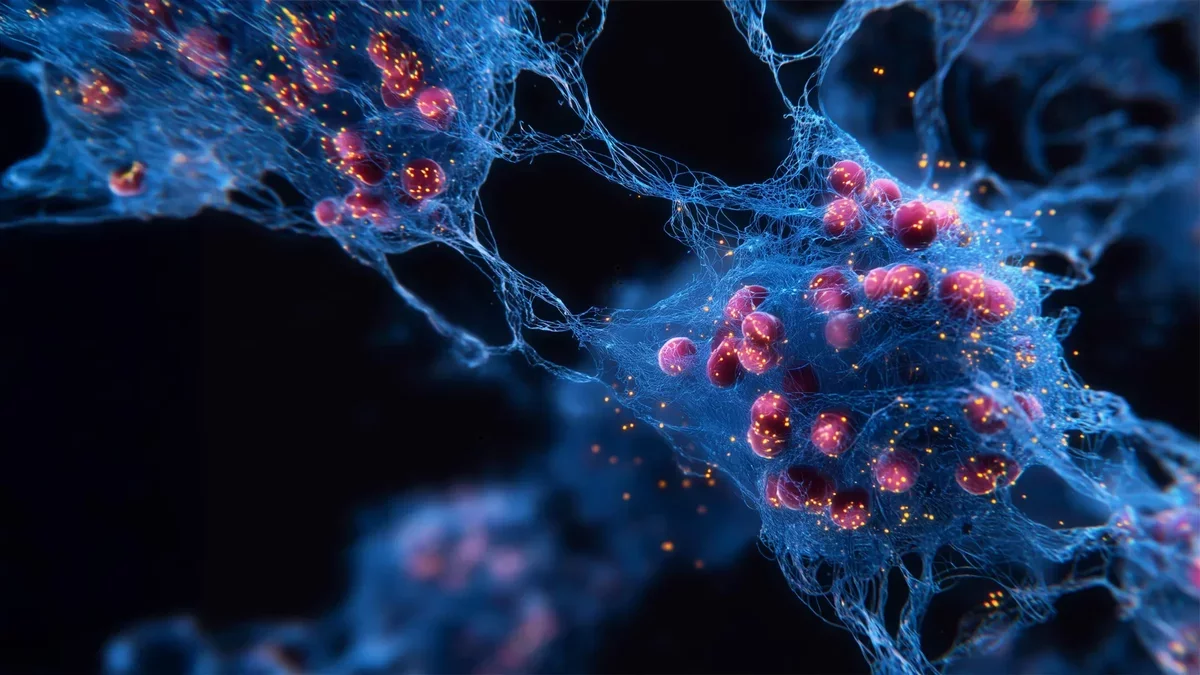
So far, the hydrogel has undergone laboratory assessments. In test tube experiments, bone-forming cells migrated into the structured hydrogel and began producing collagen, a vital component of bone. The material has been confirmed as biocompatible, posing no harm to these cells. The foundational material has been patented, and the team plans to make it available to medical manufacturers.
The ultimate goal is to introduce hydrogel-based implants for clinical use in bone repair. Further research is essential, with Qin preparing animal studies in collaboration with the AO Research Institute Davos to assess the material's effectiveness in supporting bone-forming cell movement and restoring bone strength over time.