The liver, a vital organ responsible for over 500 functions, including detoxification and blood clotting, faces significant challenges when it fails. Currently, the only viable medical solution is a full transplant, leaving over 10,000 Americans with chronic liver disease waiting for donor organs, many of whom may not survive the wait.
In a groundbreaking development, researchers at MIT have introduced an innovative alternative: injectable "mini livers." These satellite organs can be delivered directly into a patient's abdomen using a simple syringe, potentially transforming liver disease treatment.
Creating a Satellite Organ
According to Sangeeta Bhatia, a professor at MIT, these satellite livers are designed to enhance liver function while the original organ remains in place. The key to their success lies in replicating the natural environment of liver cells, known as hepatocytes, which are essential for the organ's functionality.
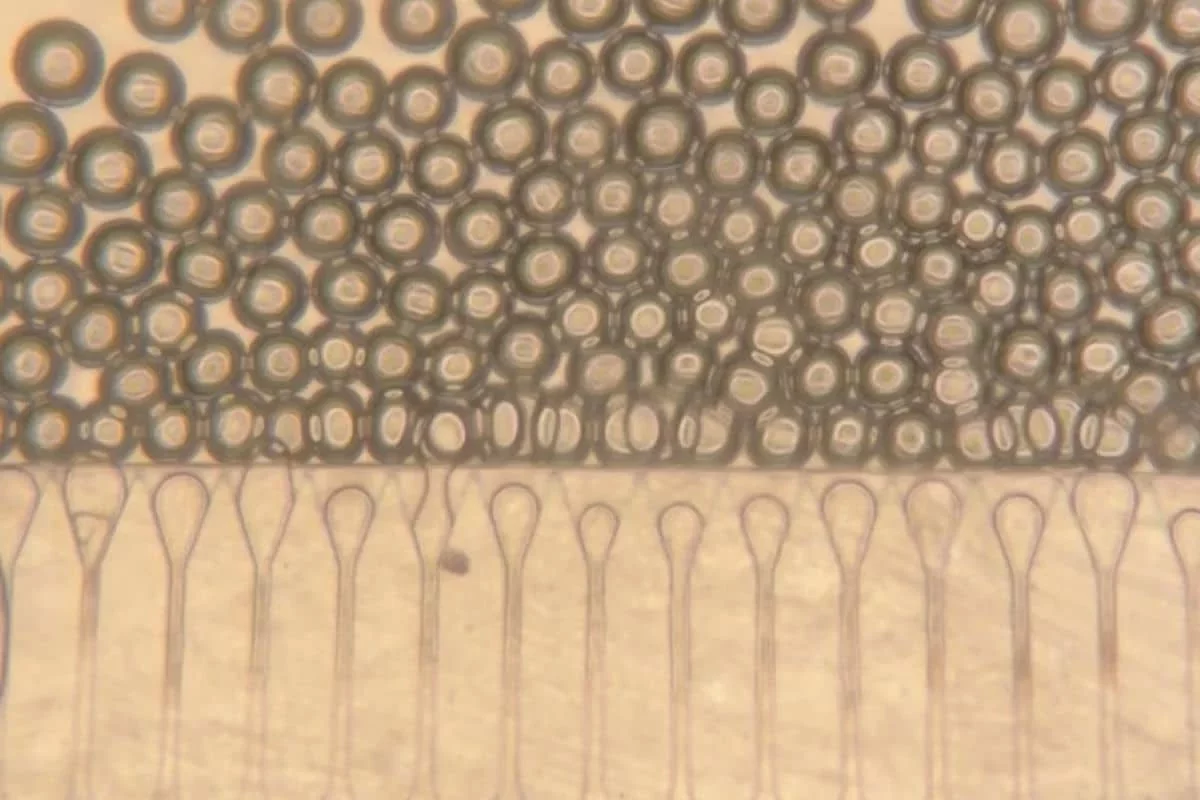
Simply injecting loose hepatocytes would lead to failure, as they would disperse without a supportive structure. To address this, MIT developed the INSITE system, which produces millions of uniform gel beads. These beads, when combined with liver cells and supportive helper cells, can be injected through a thin needle and solidify in the body, forming a stable framework for the new cells.
Targeting Belly Fat
Instead of targeting the often-damaged liver, the team focuses on a layer of abdominal fat, which provides a stable environment with ample blood supply. Using real-time ultrasound, physicians can precisely locate the injection site, as the hydrogel reflects sound waves, making it visible on monitors. This technique allows for ongoing monitoring of the satellite livers without additional incisions.
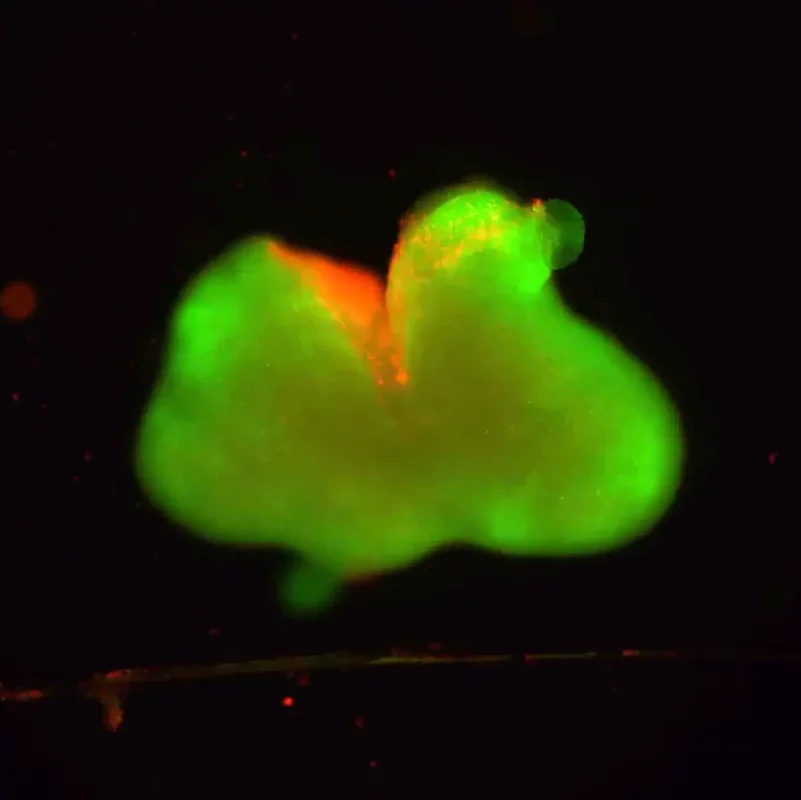
In animal studies, the results were remarkable. The host's blood vessels integrated with the new tissue, allowing the hepatocytes to thrive and perform their functions effectively, including the secretion of essential proteins into the bloodstream.
A Pathway to Future Therapies
Throughout an eight-week trial, the injected cells consistently released vital human proteins. Moreover, researchers discovered they could adjust the gel's biodegradability to control how the body remodels the implant over time.
While these findings are promising, further safety testing is essential before scaling this technology for human patients. Current protocols require patients to take immunosuppressive drugs to prevent rejection of foreign cells. To overcome this, the team is investigating the use of stem cells from the patient's own body or developing hepatocytes that can evade the immune system.
If successful, this innovative approach could revolutionize organ failure treatment. As Kumar notes, this technology not only offers a surgical alternative but also serves as a temporary solution until a donor organ is available. The researchers believe that injectable, self-assembling niches mark a significant advancement toward scalable regenerative treatments for patients awaiting organ transplants.