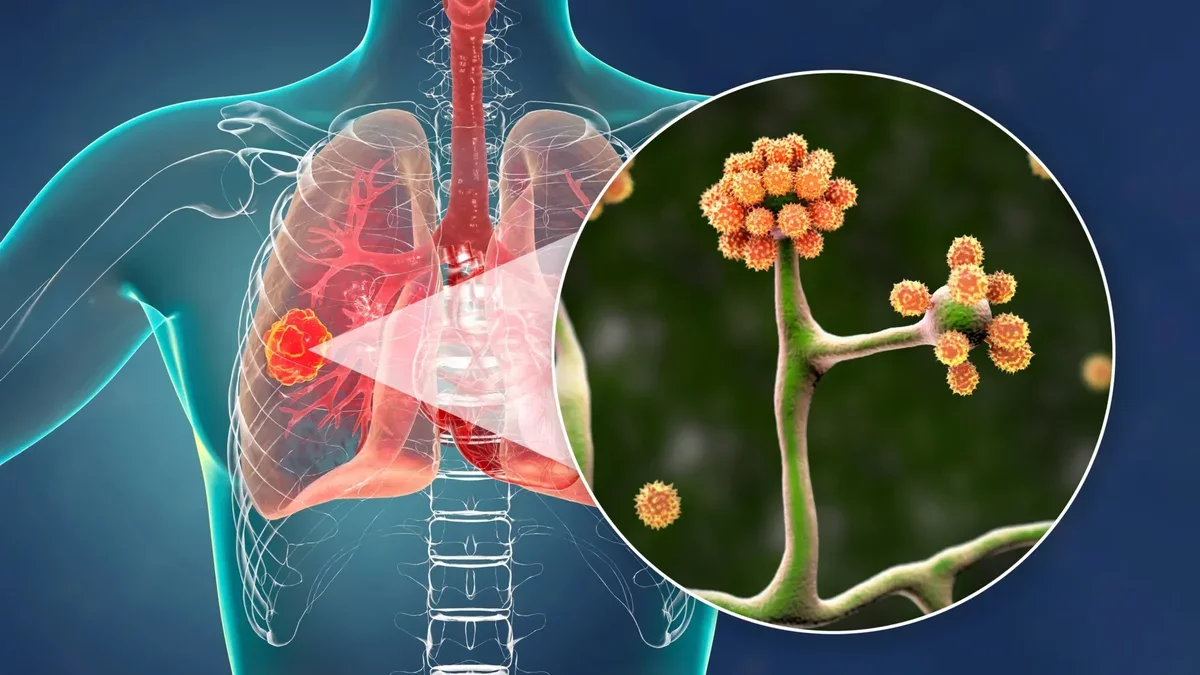
Mucormycosis, commonly known as "black fungus," is a serious infection caused by Mucorales fungi, which can rapidly disseminate throughout the body. This infection is particularly lethal, with fatality rates reaching up to 50%, especially among individuals with diabetes, weakened immune systems, or malnutrition. The incidence of mucormycosis notably increased in India during the COVID-19 pandemic.
Link Between Low Albumin Levels and Increased Mortality
Recent research indicates that patients suffering from mucormycosis exhibit significantly lower levels of albumin compared to those with other fungal infections. This condition, known as hypoalbuminemia, has been identified as a critical predictor of adverse outcomes, including death, across various patient demographics worldwide.
Dr. Ibrahim, a senior author of the study, remarked, "This discovery could transform clinical practices regarding mucormycosis." The findings suggest that hypoalbuminemia may serve as a biomarker, enabling healthcare providers to identify individuals at heightened risk for this aggressive infection. Additionally, administering albumin enriched with free fatty acids could be an effective preventive measure against mucormycosis, which is known for its rapid progression.
How Albumin Counteracts Fungal Invasion
Dr. Ibrahim elaborated on the mechanism, stating, "The study reveals how albumin neutralizes key virulence factors, including toxins and fungal proteins that contribute to tissue damage and organ invasion." The research also paves the way for potential combinations of albumin treatment with immunotherapies targeting Mucorales virulence factors, currently under development at The Lundquist Institute.
Laboratory tests demonstrated that albumin effectively inhibits the growth of Mucorales fungi without affecting other microbial populations. When albumin was absent from healthy blood samples, the fungus thrived. Furthermore, mice deficient in albumin showed increased susceptibility to infection, while restoring albumin levels provided substantial protection.
The Role of Fatty Acids
Further investigations revealed that the antifungal properties of albumin are closely linked to the fatty acids associated with the protein. These fatty acids disrupt fungal metabolism and hinder the synthesis of proteins essential for tissue invasion and disease progression. Blood samples from mucormycosis patients exhibited elevated levels of fatty acid oxidation, potentially explaining their heightened vulnerability to infection.
Collectively, these findings unveil a previously unrecognized natural defense mechanism within the human body. They also imply that therapies based on albumin could represent a groundbreaking strategy for preventing or treating mucormycosis, a condition that currently lacks effective treatment options.